
Instead of sugar doughnuts this January, why not use a lanthanide doughnut to reduce carbon dioxide?
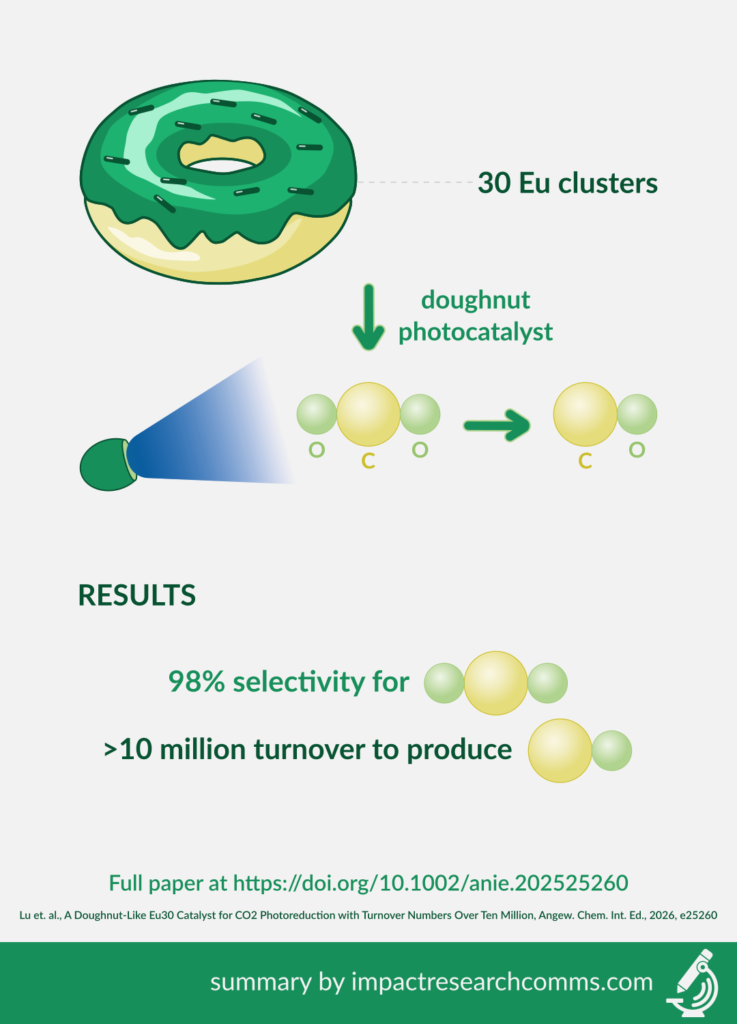
Converting carbon dioxide into useful chemicals with light can lower greenhouse gas emissions through a type of artificial photosynthesis. To accelerate this reaction, a new paper in Angewandte Chemie International Edition added a doughnut of 30 europium (Eu) clusters to transform carbon dioxide to carbon monoxide and hydrogen gas under blue LED light.
The authors created the europium doughnut in a step-by-step process, forming redox active EuIII sites for carbon dioxide binding. Adding the doughnut catalyst to a solution of acetonitrile saturated with carbon dioxide, they observed:
- 98% selectivity for CO2
- Turnover >10 million for production of CO
No carbon monoxide formed when the same doughnut structure was created from 30 dysprosium clusters, demonstrating the importance of the europium centres for the reaction.
Why does this matter?
With high selectivity and very high turnover rate, the europium-only doughnut is an effective and promising catalyst for light-based conversion of carbon dioxide to carbon monoxide.
While carbon dioxide is an environmental concern, carbon monoxide has uses in many industrial processes. This work shows that there may be a more efficient route to chemically reduce carbon dioxide into carbon monoxide with pure lanthanide photocatalysts.
Read the full paper here: https://doi.org/10.1002/anie.202525260 from Wiley
Ying Lu, Hua-Hong Zou, Jia-Wei Wang, Zhong-Hong Zhu, Fu-Pei Liang and Dongcheng Liu, A Doughnut-Like Eu30 Catalyst for CO2 Photoreduction with Turnover Numbers Over Ten Million, Angew. Chem. Int. Ed., 2026, e25260

