
Coral reefs create an outer skeleton through complex biomineralization. To build their underwater word, corals use the mineral calcium carbonate in the form of the metastable crystal aragonite.
How these skeletons grow and crystallise is shrouded in mystery. To investigate the fundamental processes of aragonite formation, a new paper in Nature Communications maps the coral skeleton surface during mineralization.
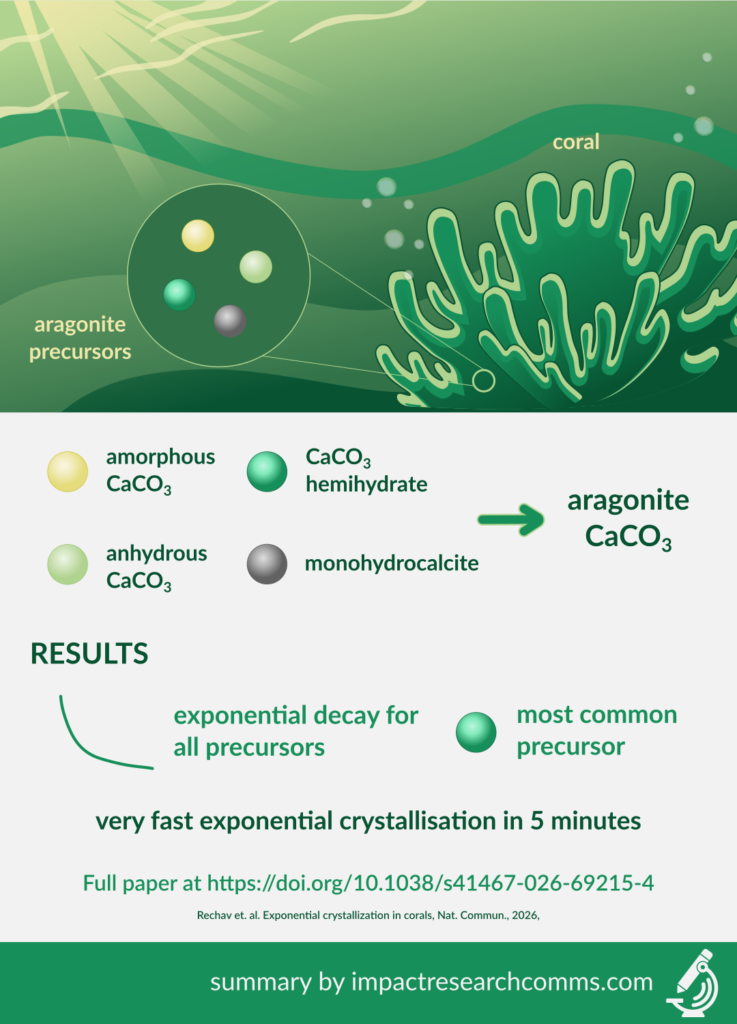
Focusing on one species of coral – Stylophora pistillata – in seawater at pH 7.2 and 8.05, the authors followed the growth from four precursor minerals:
- Amorphous calcium carbonate
- Anhydrous calcium carbonate
- Calcium carbonate hemihydrate
- Monohydrocalcite
All precursors exponentially decayed, with the most dominant being the crystalline precursor 3, to form aragonite. The crystallisation rate for all phases was very fast, occurring in 5 minutes. Overall, aragonite formed following exponential kinetics, regardless of precursor, time, and pH, and continued to form even 6 months after death.
Why does this matter?
Observing the very early stages of crystallisation is challenging, especially in living organisms. Understanding how minerals form in complex, natural systems is important for protecting natural history from biological and geological sources, while also informing the future development of synthetic materials.
Read the full paper here: https://doi.org/10.1038/s41467-026-69215-4 from Springer Nature
Zoë Rechav, Eric Tambutté, Isabelle M. LeCloux, Samantha Anglemyer, Natalie E. Beltz, Nicolas A. Chou, Brynne E. Dixson-Kruijf, Johannes Domagk, Anders M. Larson, Sylvia W. Lewis, Rhita Rich, Lateef O. Saheed, James L. Schwenk, Jaden S. Sengkhammee, Christian A. Waltenberg, Jianfeng Ye, Barat Q. Achinuq, Alexander A. Venn, Sylvie Tambutté and Pupa U. P. A. Gilbert, Exponential crystallization in corals, Nat. Commun., 2026

